Welcome from the Computational Modelling Group

Welcome to the website of the CoMo Group. We develop and apply modern numerical methods to problems arising in Chemical Engineering. The overall aim is to shorten the development period from research bench to the industrial production stage by providing insight into the underlying physics and supporting the scale-up of processes to industrial level.
The group currently consists of 15 members from various backgrounds. We are keen to collaborate with people from both within industry and academia, so please get in touch if you think you have common interests.
The group's research divides naturally into two inter-related branches. The first of these is research into mathematical methods, which consists of the development of stochastic particle methods, computational fluid dynamics and quantum chemistry. The other branch consists of research into applications, using the methods we have developed in addition to well established techniques. The main application areas are reactive flow, combustion, engine modelling, extraction, nano particle synthesis and dynamics. This research is sponsored on various levels by the UK, EU, and industry.
Recent News  Subscribe
Subscribe
Prof. Kraft shares vision of Human-in-the-Loop
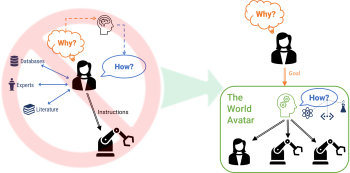
In a recent article for the Data-Centric Engineering Blog on Medium, Prof. Kraft discusses an impending paradigm shift in interactions between humans and machines enabled by consistent knowledge models. He envisions humans to define overarching goals based on which intelligent systems derive sub-goals and create a roadmap for attaining the goals.
The CoMo group hopes to facilitate this shift by developing a common framework for knowledge representation and discovery in The World Avatar project. This is reflected in our recent work in the field of laboratory automation. We explain key ideas behind our view of Human-in-the-Loop systems along with design principles and technical details in a new white paper.
Congratulations to Jiying Chen for Winning Best Poster Prize at 2023 CEB Annual Research Conference

A CoMo poster has been awarded the Best Poster Prize in the Sustainability Session at the 2023 CEB Annual Research Conference, showcasing a knowledge graph-based approach for geospatial data integration.
We are pleased to share the news of the recognition received by our 2nd-year PhD student, Jiying Chen, at the 2023 CEB Annual Research Conference. His poster, "Advancing Sustainable Smart Cities: A Knowledge Graph-based Approach for Data Integration," has been acknowledged for its thoughtful approach to data integration, with a specific focus on sustainable smart cities. This recognition highlights the efforts made by Chen and his colleagues, underlining the substantial contributions we are making to sustainable cities and knowledge modelling. We look forward to seeing the continued positive impact of this research.
Prof. Kraft's Discussion on "The World Avatar Project" with ChemistryViews

We are excited to share that Prof. Kraft has been featured in an engaging conversation with Dr. Vera Koester on ChemistryViews. Central to this discussion is his research initiative, "The World Avatar (TWA) project". This pioneering work is centred on establishing an interconnected network of digital twins with the potential to detail the operations of multifaceted systems. Such advancements are poised to streamline data-driven decisions, encompassing a range of applications, from chemical knowledge synthesis and laboratory automation to enhancing smart city operations, bolstering climate resilience, and shaping national energy landscapes. In the discussion, Prof. Kraft provides clarity on the fundamentals of knowledge engineering and machine learning, emphasizing their significant roles in modern chemistry. Moreover, he delves into the most pressing challenges and rewarding successes encountered in the domain of Artificial Intelligence.
Account Article on Knowledge Engineering in Chemistry

The CoMo group is excited to announce its latest account article published in Acc. Chem. Res., titled "Knowledge Engineering in Chemistry: From Expert Systems to Agents of Creation". Penned by Aleksandar Kondinski, Jiaru Bai, Sebastian Mosbach, Jethro Akroyd, and Markus Kraft, the article delves into the evolution of knowledge engineering in the chemical domain. It highlights the progression from early expert systems to today's advanced tools, including knowledge graphs and cognitive agents. Furthermore, the narrative introduces the innovative concept of The World Avatar (TWA) within chemical research.
The World Avatar added to WEF’s list of Global Use Cases
The World Avatar project has been selected for the World Economic Forum (WEF)’s list of Global Use Cases as part of its Global Digital Twin Cities initiative.
The initiative offers a potential solution for policymakers worldwide to improve city governance and create a conducive urban ecosystem for industries and people. It is a collaboration between the WEF’s Urban Transformation Platform, The China Academy of Information and Communication Technology (CAICT), and other key stakeholders.
At the beginning of 2021, the WEF and the CAICT launched the three-year initiative to make digital twin technology accessible to the public, promote social equity and inclusiveness, help cities achieve digital and low-carbon transformation, and jointly shape the future of digital twin city development. Ultimately, WEF hopes to render cities more inclusive, sustainable, and liveable through these efforts.
The World Avatar and the WEF
The World Avatar (TWA) project perfectly fits as an example for this WEF initiative. TWA represents the digital world using a dynamic knowledge graph containing concepts and data describing the world. TWA simulates the world’s behaviour with an ecosystem of autonomous computational agents that update the concepts and data so that the digital world remains current in time.
In this manner, it is possible to create digital twins that can describe the behaviour of any complex system and can make data-driven decisions about how to optimise the systems. TWA can improve interoperability across all sectors by providing a solution that enables the integration of distributed heterogeneous models and data. By including a description of the computational agents within the TWA knowledge graph, we can confer the discoverability of the computational capabilities and easily combine heterogeneous pieces of software and data.
Uses of The World Avatar
TWA contains a description of the UK’s electrical power and gas transmission systems. This gives geospatial details of infrastructure and captures relationships between different infrastructure elements and live data feeds describing gas flow into the transmission system. TWA can leverage disparate information from gas transmission systems, gas consumption data, and climate observations to investigate the hypothetical outcome of switching from gas heating to heat pumps.
Using TWA, it was discovered that while a complete switch to heat pumps would reduce the UK’s total carbon dioxide emissions by approximately 10%, it would exacerbate fuel poverty in colder parts of the country. In this way, TWA allows the UK government to examine specific scenarios in enough detail to support local policymaking.
TWA can also contain descriptions of the geometry and features of buildings, including details of individual apartments and the contents of individual rooms. Once the buildings are described in TWA, they can be processed by computational agents acting on the knowledge graph.
For example, TWA has a representation of the CARES lab in Singapore. Data about the building, the laboratory layout, and the locations of sensors in the laboratory are all represented as part of TWA’s knowledge graph. Agents then use real-time data about the ambient humidity and outdoor air temperature to achieve a specified temperature in the lab, and flammable chemicals can be monitored by incorporating sensors into the safety cabinet in which they are stored.
Another fitting application of TWA is the Climate Resilience Demonstrator (CReDo), developed by CARES’ collaborator CMCL Innovations as part of the UK’s National Digital Twin programme. CMCL applied ideas from TWA to create a digital twin that integrated data describing assets from the water, energy, and telecom industries. Data from flood simulations were then combined with asset data to model the potential cascade of failures through the combined asset network in future flood scenarios for cities.
TWA is much more than just these examples. With its dynamic knowledge graph technology and ecosystem of autonomous computational agents, it can create digital twins that can describe the behaviour of any complex system and can make data-driven decisions about how to optimise the systems. WEF’s Global Digital Twin Cities initiative has recognised the value of this approach, and by adding CARES and TWA to its Global Use Cases, it stands as an example of how to reach true interoperability between diverse sectors.
New paper - Soot inception: Carbonaceous nanoparticle formation in flames
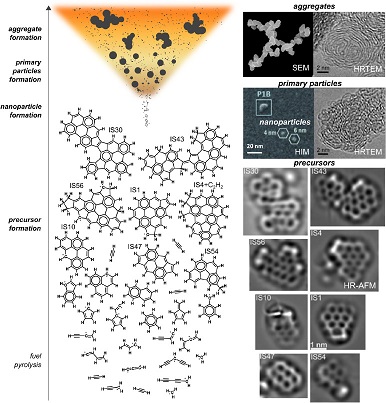
Our new review on soot inception was just published in Progress in Energy and Combustion Science
Abstract
The route by which gas-phase molecules in hydrocarbon flames form condensed-phase carbonaceous nanoparticles (incipient soot) is reviewed. These products of incomplete combustion are introduced as particulates and materials revealing both their useful applications and unwanted impacts as pollutants. Significant advances in experimental techniques in the last decade have allowed the gas phase precursors and the transformation from molecules to nanoparticles to be directly observed. These measurements combined with computational techniques allow for various mechanisms known to date to be compared and explored. Questions remain surrounding the various mechanisms that lead to nanoparticle formation. Mechanisms combining physical and chemical routes, so-called physically stabilised soot inception, are highlighted as a possible “middle way”.
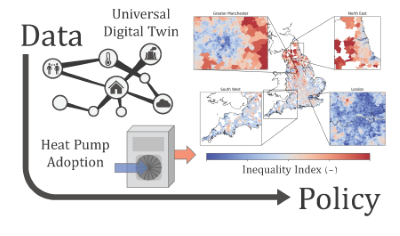
Will using heat pumps for domestic heating increase or decrease inequality? We use the Universal Digital Twin to investigate...
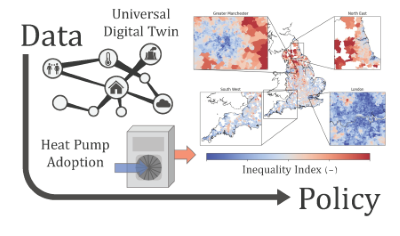
Preprint 281, ''Universal Digital Twin – the impact of heat
pumps on inequality'', has been published!
Abstract
This paper investigates how using heat pumps for domestic heating would impact fuel poverty and inequality. The analysis integrates a geospatial description of climate observations, gas and electricity infrastructure, energy consumption and fuel poverty from the base world of a Universal Digital Twin based on the World Avatar knowledge graph. Historic temperature data were used to estimate the temporal and geospatial variation of the performance of air source heat pumps in the UK. The corresponding change in gas and electricity consumption that could be achieved using heat pumps instead of gas for domestic heating was estimated. The geospatial impact of the heat pumps was assessed in terms of CO2 savings, and their effect on fuel cost and fuel poverty. Whilst heat pumps would reduce emissions, it is predicted that they would increase fuel costs. It was shown that both local and regional areas of high fuel poverty would experience some of the largest increases in fuel cost. This illustrates the potential for the transition to sustainable heating to exacerbate inequality. The analysis suggests that existing regional inequalities will increase, and that it comes down to a political choice between investments to support the most effective use of heat pumps, and delayed investments to counter inequality. The ability of the World Avatar to integrate the models and data necessary to perform this type of analysis provides a means to generate actionable information, for example, to enable local policy interventions to address the tension between social and environmental goals.
CoMo group open to Feodor Lynen Research Fellows

In 2016, Prof. Markus Kraft was awarded the Friedrich Wilhelm Bessel Award and is therefore eligible to host Feodor Lynen Research Fellows sponsored by the Alexander von Humboldt Foundation. The Feodor Lynen Research Fellowship covers the salary and travel expenses of researchers from Germany to work at the host institution for 6-24 months. In addition, the fellowship enables the successful candidate to apply for alumni sponsorship from the Humboldt Foundation after the end of the fellowship and become part of their international network of academics.
If you are interested in working at the University of Cambridge and in joining the CoMo group as a post-doctoral researcher, please check your eligibility on the official Feodor Lynen Research Fellowship website and familiarise yourself with the application procedure. You will need to write a research proposal that aligns with your professional expertise. The topic might be of computational or experimental nature but should lie within the research areas of the CoMo group.